SOLVED: A solution made by mixing 569 mL of water and 238 mL ethanol: What is the concentration of ethanol in units of volume/volume percent? Please explain briefly the reason why YOU
The relationship between SPM concentration by mass and volume (a) and... | Download Scientific Diagram

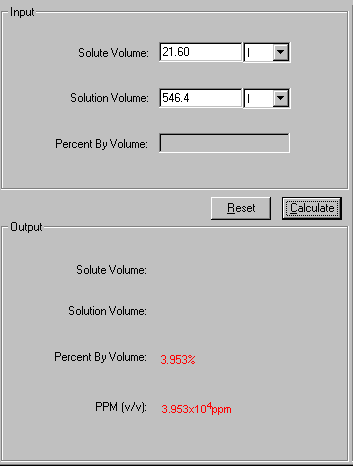
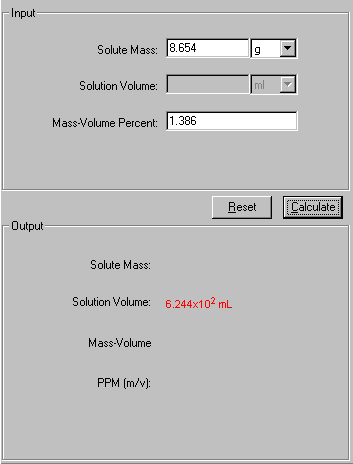
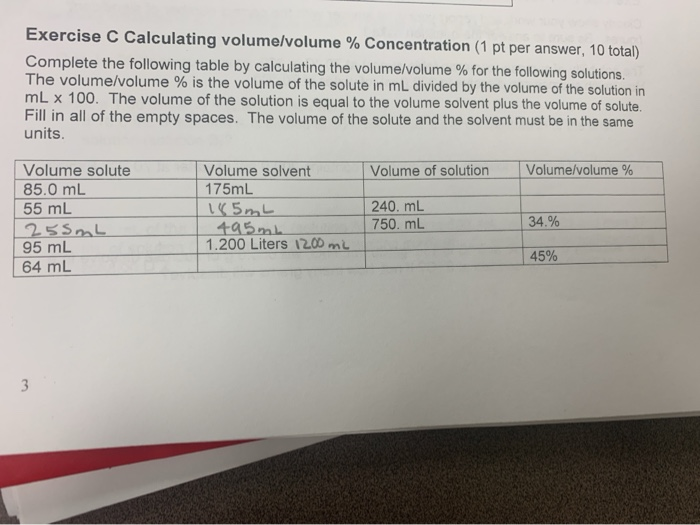
Concentrations and Consumer Products. Percentage Volume/Volume (% v/v) Commonly used when both the solute and the solvent are liquids volume/volume = - ppt download




:max_bytes(150000):strip_icc()/GettyImages-493151728-c08e9b2d60bb401b8bbdd2e66de7a93b.jpg)