The volume of a drop of water is `0.04 mL`. How many `H_(2) O` molecules are there in a drop of a water? `d = 1.0 g mL`.

SIP IS (1) 2.7 x 1019 (2) 1.08 x 1020 (3) 10.8 x 1020 (4) 5.4 x 1019 The volume of a drop of water is 0.05 ml and the density of
Calculating the Volume of Water Droplets on Topographically-Modified, Micro-Grooved Aluminum Surfaces

The volume of a drop of water is 0.05 ml and the density of water is 1.0 g/ml. How many water molecules are present in a drop of water?

S CAREER INSTITUTE 46. The volume of a drop of water is 0.0018 mL then the number of water molecules present in two drop of water room temperature is :- (1) 12.046

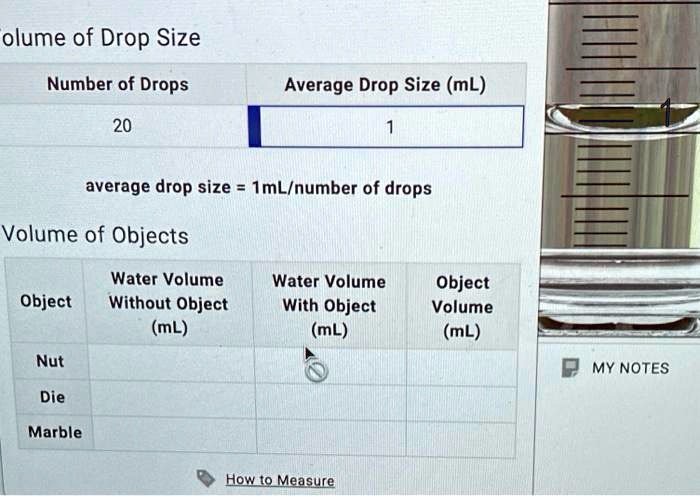
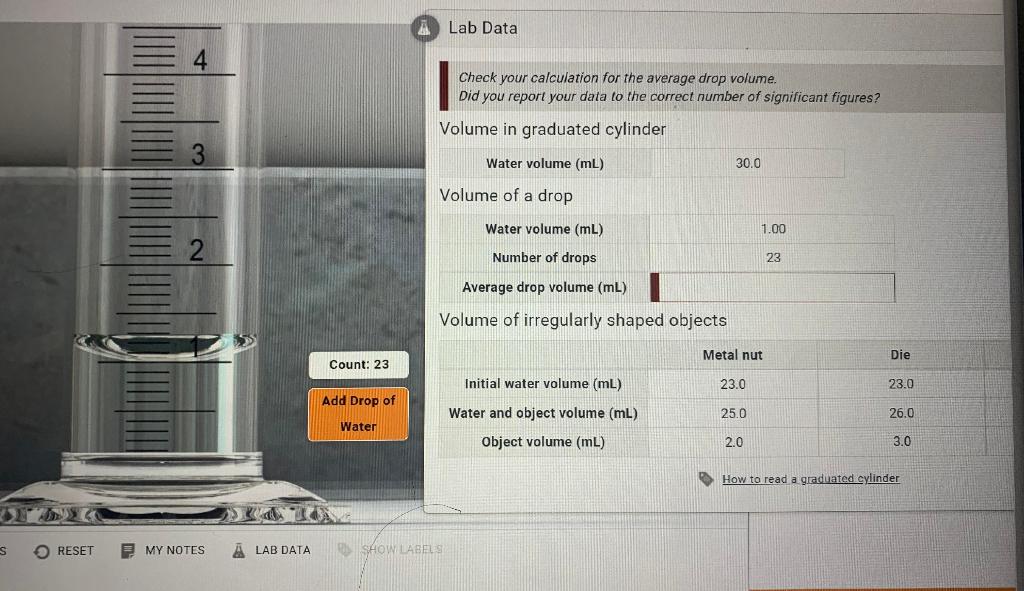
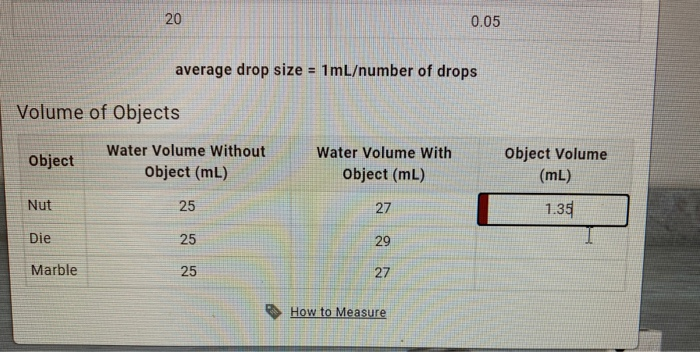
SOLVED: Lab Data Liquid Level Measurement Water Volume 030 000 mL = Liter Volume of Drop Size Number of Drops Average Drop Size (mL) 0.05 average drop size (mL/number of drops) Volume