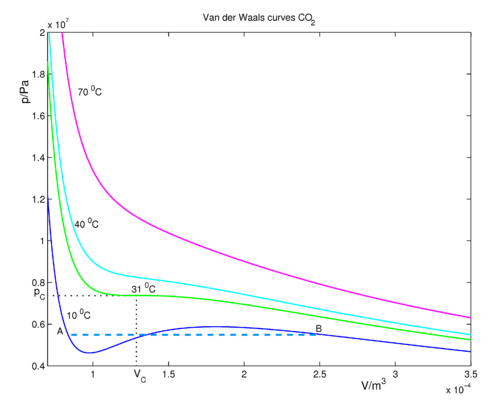
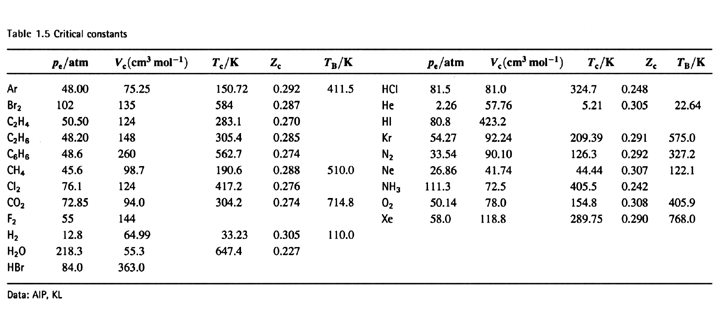
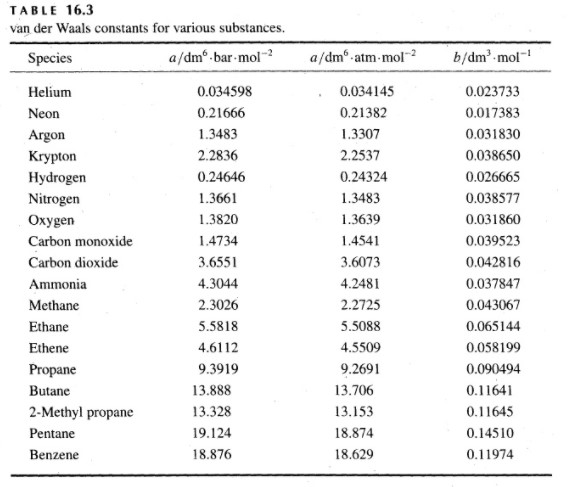
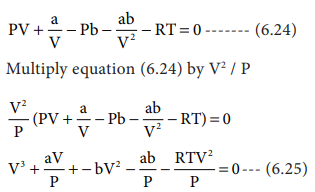physical chemistry - Why does small value of van der Waals gas constant "b" ensure easier liquefication? - Chemistry Stack Exchange

ReasonThe gases obeying van der Waal's equation in all the conditions of pressure and temperature are called van der Waal's gases.AssertionFor a van der Waal's gas, the constants 'a' and 'b' are