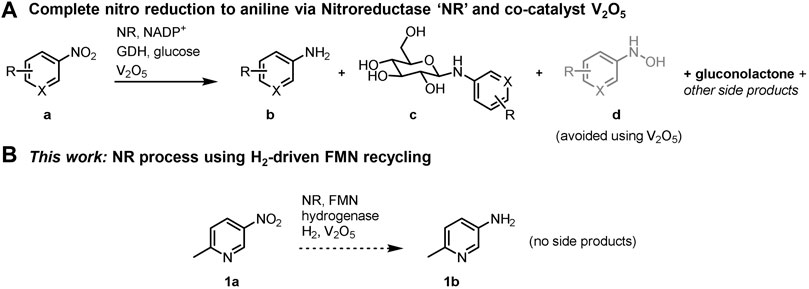
Frontiers | H2-Driven Reduction of Flavin by Hydrogenase Enables Cleaner Operation of Nitroreductases for Nitro-Group to Amine Reductions

Reduction of nitrobenzene in acidic media in presence of a reducing... | Download Scientific Diagram

The mechanism of reduction of a) three nitro groups in the most likely... | Download Scientific Diagram
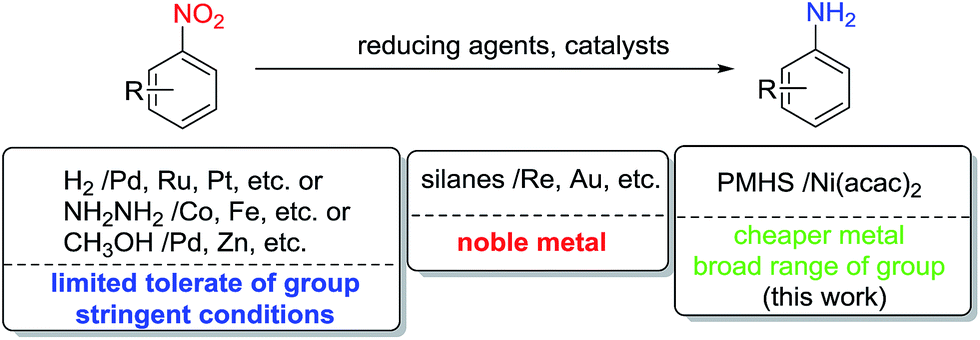
Selective reduction of nitro-compounds to primary amines by nickel-catalyzed hydrosilylative reduction - RSC Advances (RSC Publishing) DOI:10.1039/C5RA17731F

Chemoselective nitro reduction and hydroamination using a single iron catalyst. - Abstract - Europe PMC
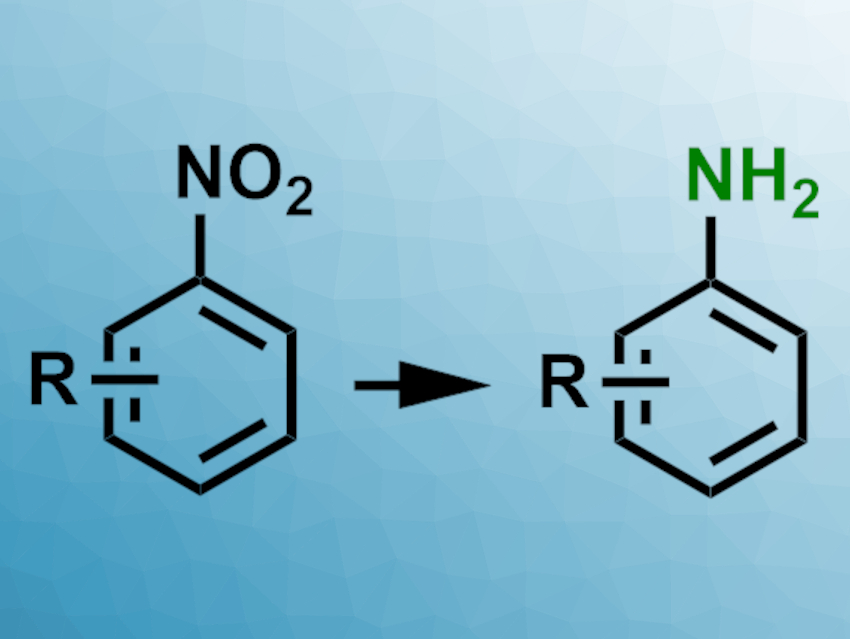
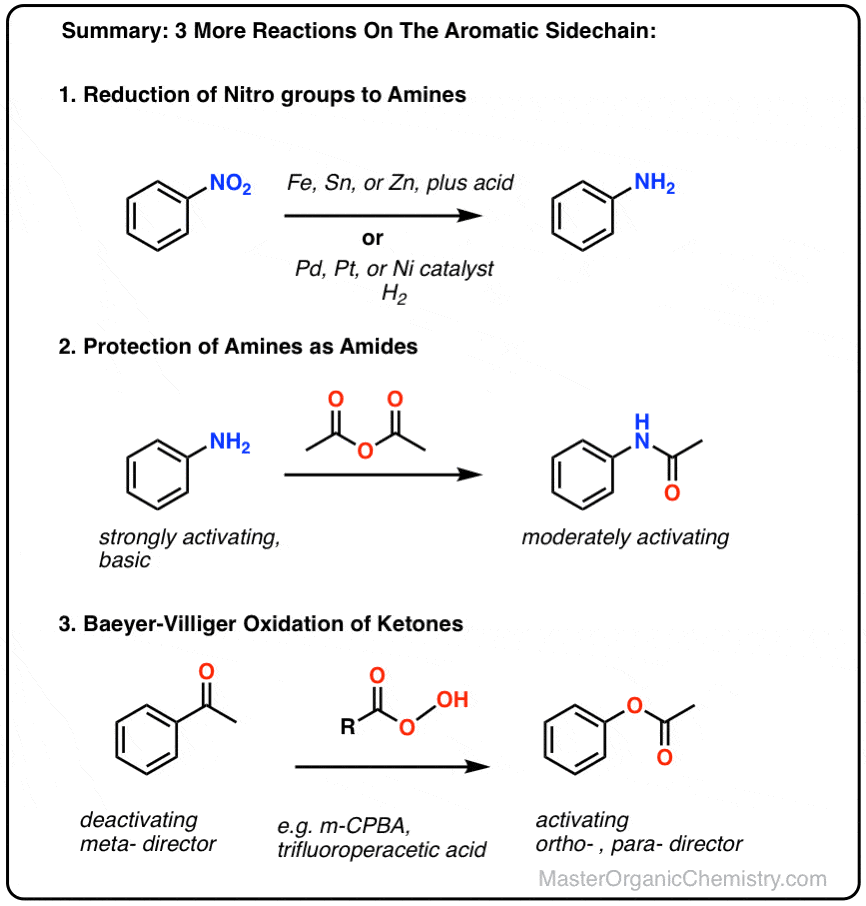
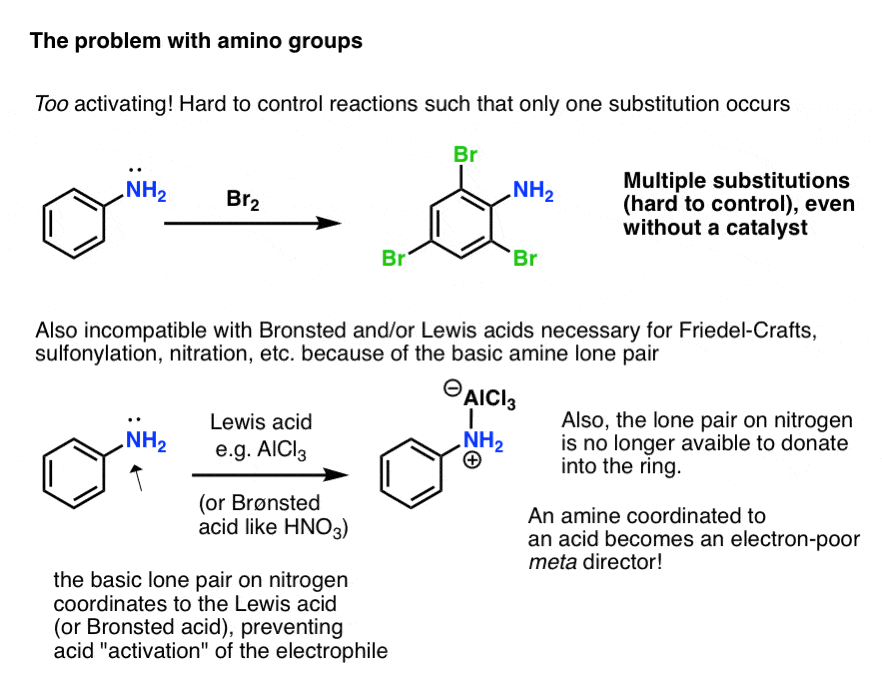
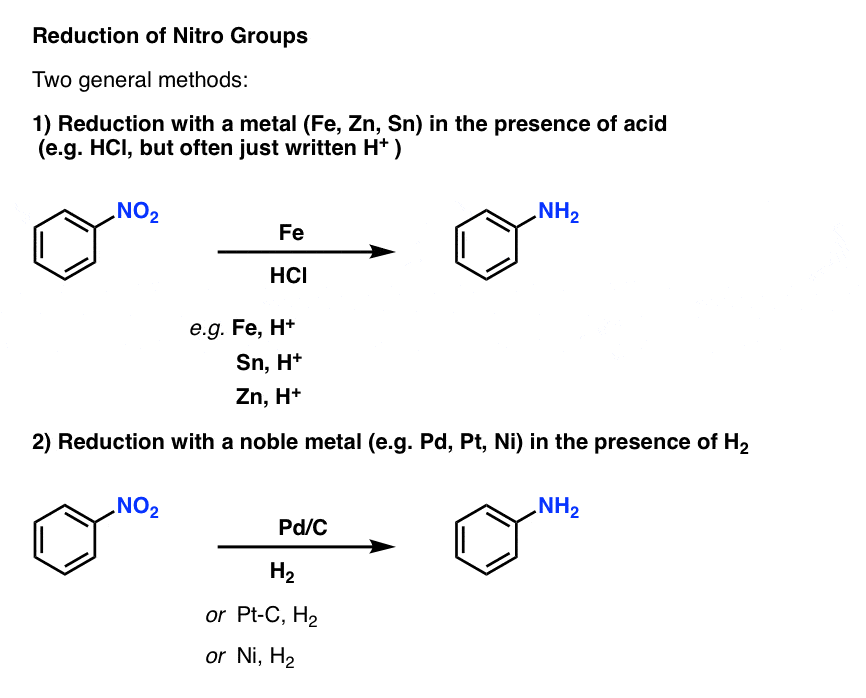
38 Which of the following reagents would not be a good choicefor reducing anaryl nitro compound to an amine? (1) H2(excess)/Pt (2) LiAlH4 in ether. (3) Fe and HCl (4) Sn and HCl