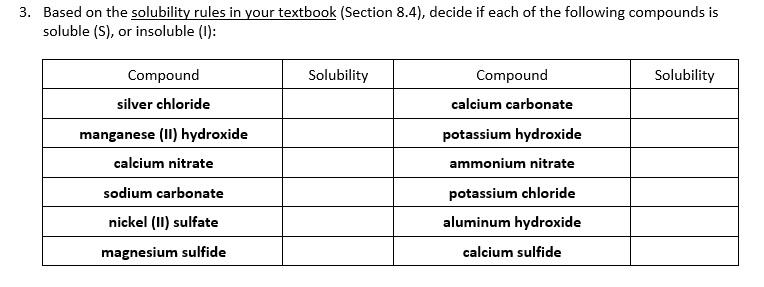
TGA-DSC curves of Mn(NO 3 ) 2 $ 4H 2 O decomposition in air (heating... | Download Scientific Diagram

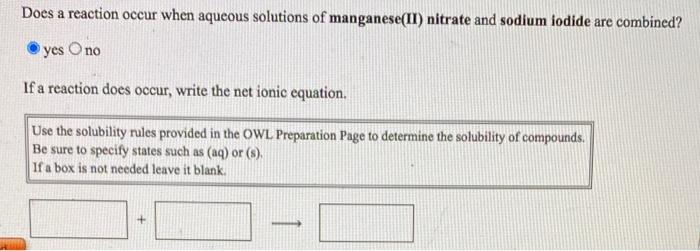
SOLVED: Use the solubility generalizations on the information page to predict if one or more precipitates will form when aqueous solutions of manganese(II) sulfate (MnSO4) and zinc nitrate (Zn(NO3)2) are mixed. Write
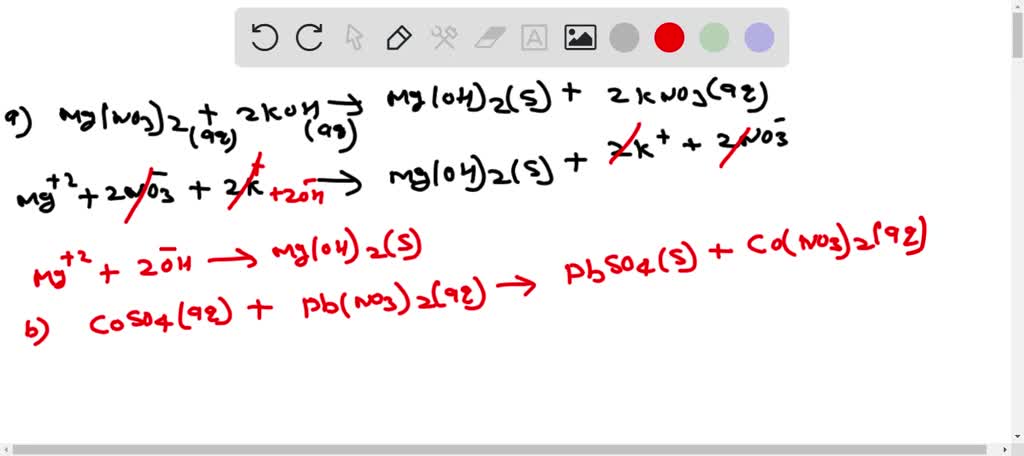
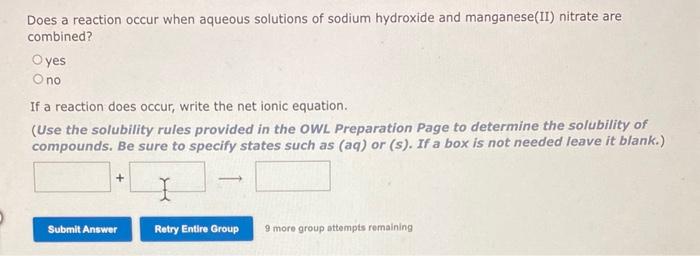
SOLVED: Does a reaction occur when aqueous solutions of manganese(II) nitrate and potassium hydroxide are combined? yes no If a reaction does occur, write the net ionic equation. Use the solubility rules
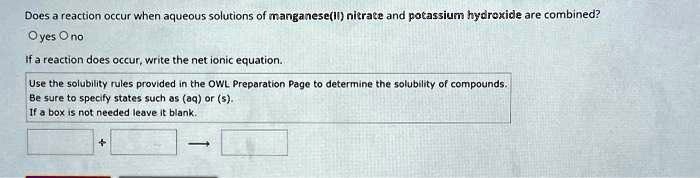
SOLVED: Does a reaction occur when aqueous solutions of manganese(II) nitrate and potassium hydroxide are combined? Yes/No. If a reaction does occur, write the net ionic equation. Use the solubility rules provided

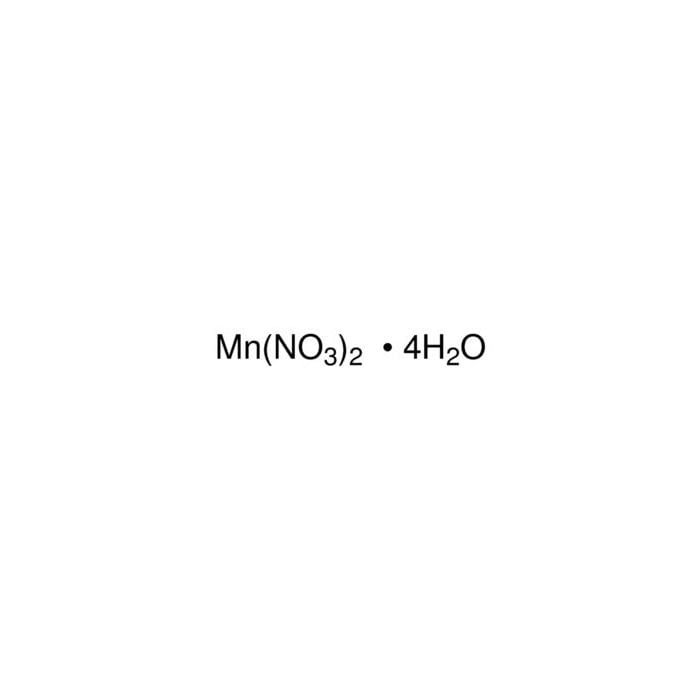
Solubility of manganese nitrate hexahydrate in water between 250 and 300 K | Download Scientific Diagram

VIDEO solution: When solubility rules are applied, manganese solid (Mn) nitrate solution is mixed with a potassium sulfide solution under the right conditions. According to the correct balanced equation for the formation

SOLVED: The compound manganese(II) nitrate, Mn(NO3)2 is soluble in water. Write the net ionic equation for the dissociation reaction that occurs when solid manganese(II) nitrate dissolves in water: Use the pull-down boxes