DBU‐Promoted Intramolecular Crossed Aldol Reaction: A Facile Access to Indane‐Fused Pyrrolidine - Yang - 2019 - European Journal of Organic Chemistry - Wiley Online Library

10 .Csir net December 2017 organic chemistry solution Iodo lactonization,DBU reagent in hindi - YouTube

NBS/DBU-Promoted One-Pot Three-Component Cycloaddition of Malonic Acid Derivatives, Nitrosoarenes, and Alkenes: Synthesis of Isoxazolidines | The Journal of Organic Chemistry
Proposed mechanisms for the DBU catalyzed urea-synthesis reaction from... | Download Scientific Diagram
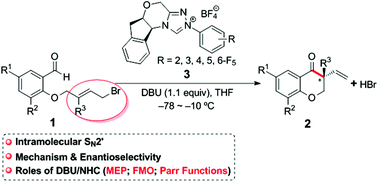
Theoretical study on the mechanism and enantioselectivity of NHC-catalyzed intramolecular SN2′ nucleophilic substitution: what are the roles of NHC and DBU? - Organic Chemistry Frontiers (RSC Publishing)

NBS/DBU-Promoted One-Pot Three-Component Cycloaddition of Malonic Acid Derivatives, Nitrosoarenes, and Alkenes: Synthesis of Isoxazolidines - ScienceDirect
![College: Organic Chemistry] Does this mechanism make sense, the last step is confirmed in a paper but i'm trying to figure out the DBU catalysed mechanism (first step) : r/chemistryhomework College: Organic Chemistry] Does this mechanism make sense, the last step is confirmed in a paper but i'm trying to figure out the DBU catalysed mechanism (first step) : r/chemistryhomework](https://i.redd.it/t5hrthkdks821.jpg)
College: Organic Chemistry] Does this mechanism make sense, the last step is confirmed in a paper but i'm trying to figure out the DBU catalysed mechanism (first step) : r/chemistryhomework
![Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/da071cf8-eee0-4e2f-8e22-5b6f6a0f91e7/adsc201601279-fig-5004-m.jpg)
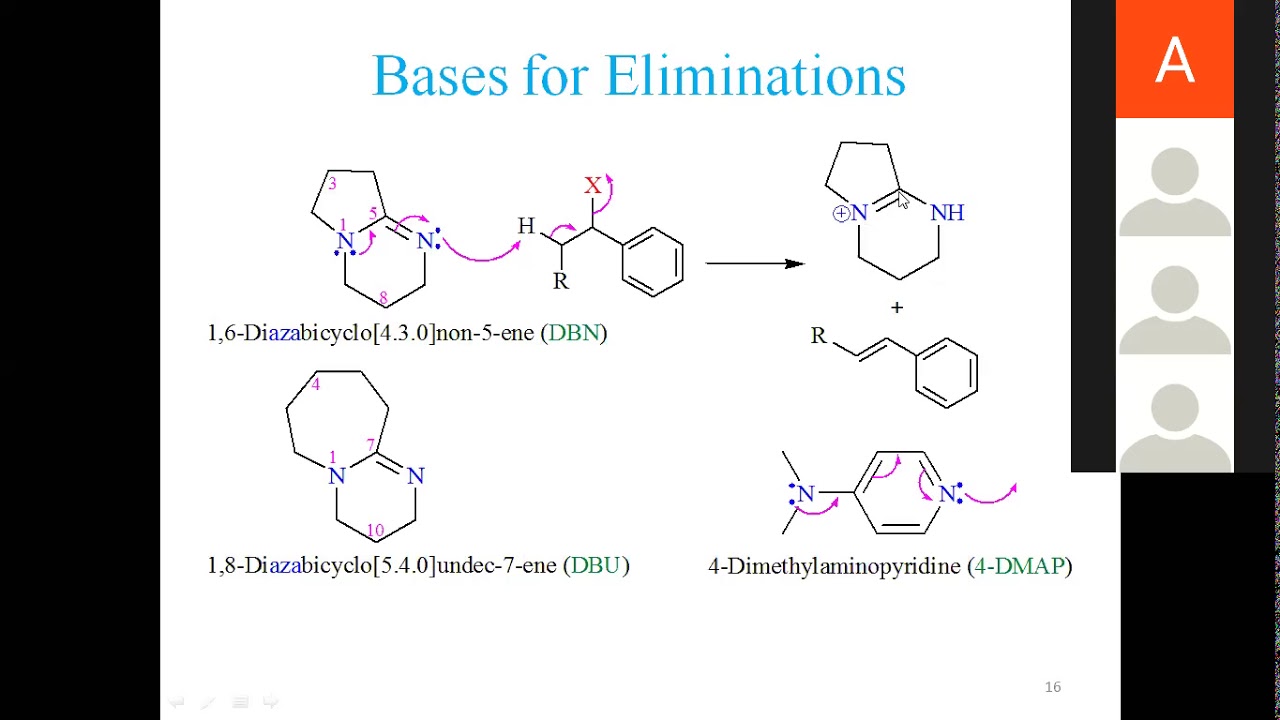
Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library
![The Reaction of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) with Carbon Dioxide | The Journal of Organic Chemistry The Reaction of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) with Carbon Dioxide | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo0503759/asset/images/jo0503759.social.jpeg_v03)
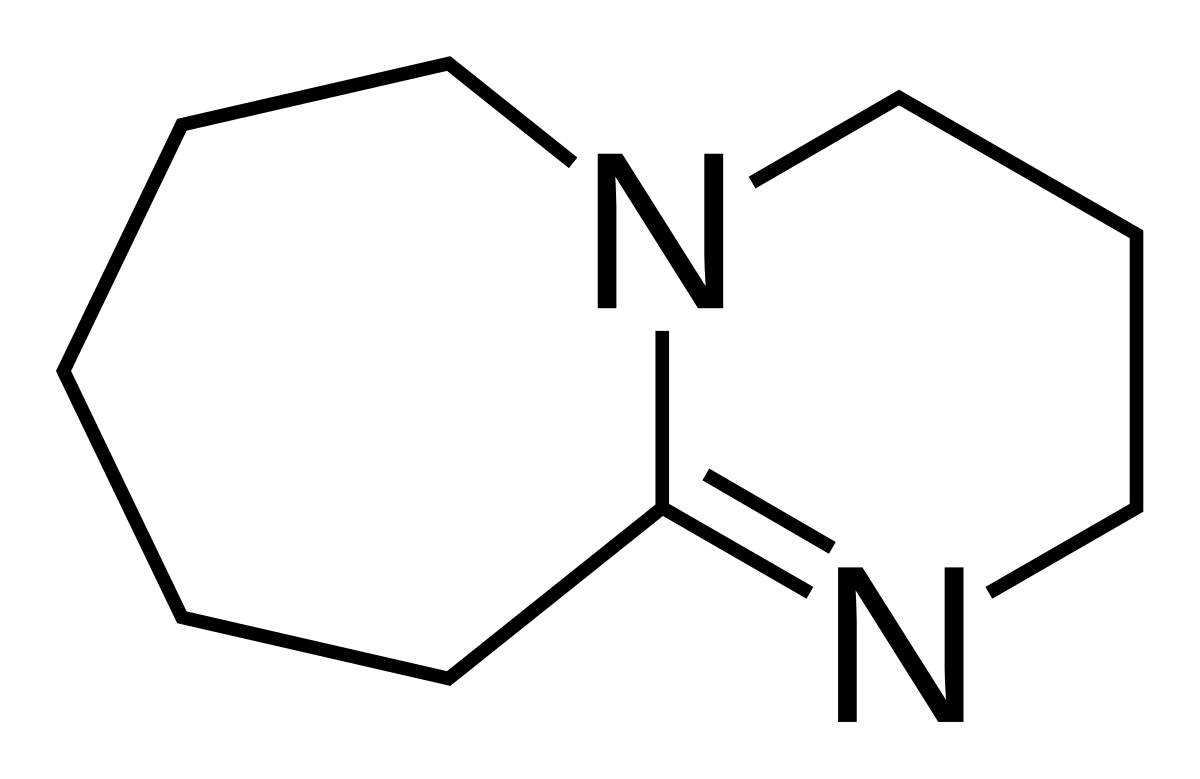
The Reaction of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) with Carbon Dioxide | The Journal of Organic Chemistry
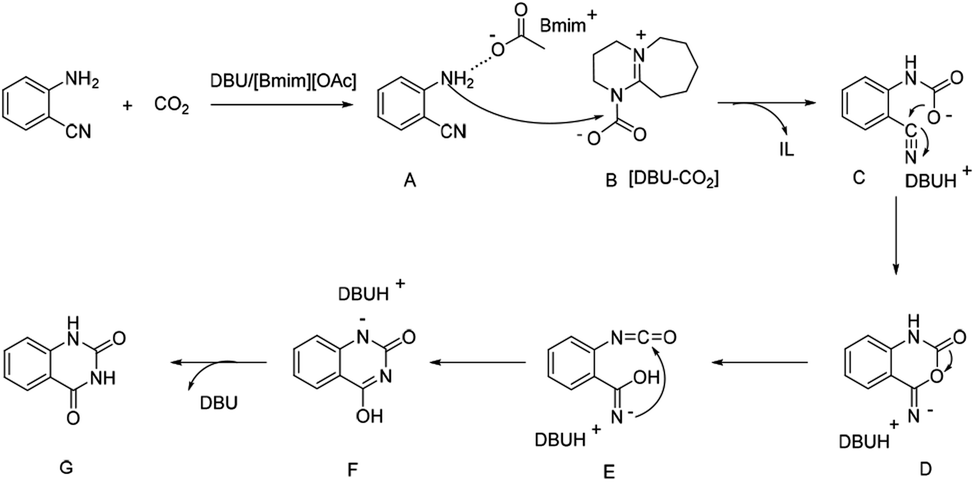
DBU coupled ionic liquid-catalyzed efficient synthesis of quinazolinones from CO 2 and 2-aminobenzonitriles under mild conditions - RSC Advances (RSC Publishing) DOI:10.1039/D0RA00194E
![The Dual Role of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) in the Synthesis of Terminal Aryl- and Styryl-Acetylenes via Umpolung Reactivity | Organic Letters The Dual Role of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) in the Synthesis of Terminal Aryl- and Styryl-Acetylenes via Umpolung Reactivity | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.5b02398/asset/images/large/ol-2015-02398j_0006.jpeg)
The Dual Role of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) in the Synthesis of Terminal Aryl- and Styryl-Acetylenes via Umpolung Reactivity | Organic Letters
![1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU): A Versatile Reagent in Organic Synthesis | Bentham Science 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU): A Versatile Reagent in Organic Synthesis | Bentham Science](https://www.eurekaselect.com/images/graphical-abstract/coc/19/9/0005D.gif)
1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU): A Versatile Reagent in Organic Synthesis | Bentham Science
![Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences](https://royalsocietypublishing.org/cms/asset/0ea92727-dfe5-4dad-b4fd-c68f2d8d3d5e/rspa20190238f03.jpg)
Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences

Four-Component Cyclization of Naphthol/Thionaphthol/Naphthylamine, Formaldehyde, and DBU in Water | The Journal of Organic Chemistry

![1,8-Diazabicyclo[5.4.0]undec-7-ene, 98 %, Thermo Scientific Chemicals | Fisher Scientific 1,8-Diazabicyclo[5.4.0]undec-7-ene, 98 %, Thermo Scientific Chemicals | Fisher Scientific](https://assets.fishersci.com/TFS-Assets/CCG/Chemical-Structures/chemical-structure-cas-6674-22-2.jpg-650.jpg)


![DBU [1,8-Diazabicyclo[5,4,0]undec-7-ene] - An Overview - YouTube DBU [1,8-Diazabicyclo[5,4,0]undec-7-ene] - An Overview - YouTube](https://i.ytimg.com/vi/fu67cE_ydew/maxresdefault.jpg)