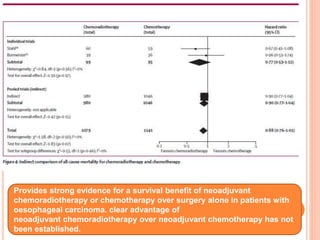
Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial - ScienceDirect

Outcome of Patients Treated Within and Outside a Randomized Clinical Trial on Neoadjuvant Chemoradiotherapy Plus Surgery for Esophageal Cancer: Extrapolation of a Randomized Clinical Trial (CROSS) | Annals of Surgical Oncology
![PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/630bc6b873bf8304e24a6ac1260e289c3deac5d2/8-Figure2-1.png)
PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar

integration NEOPEC-trial within CROSS-trial. CRT chemoradiotherapy, PET... | Download Scientific Diagram
Survival Between Neoadjuvant and Definitive Chemoradiotherapy In Locally Advanced Esophageal Cancer | Chaochankit | Journal of Gastroenterology and Hepatology Research

CRITICS-II: a multicentre randomised phase II trial of neo-adjuvant chemotherapy followed by surgery versus neo-adjuvant chemotherapy and subsequent chemoradiotherapy followed by surgery versus neo-adjuvant chemoradiotherapy followed by surgery in ...
![PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/630bc6b873bf8304e24a6ac1260e289c3deac5d2/5-Figure1-1.png)
PDF] Accuracy of Detecting Residual Disease After Cross Neoadjuvant Chemoradiotherapy for Esophageal Cancer (preSANO Trial): Rationale and Protocol | Semantic Scholar

PDF) 272 10-YEAR Follow-Up of a Randomised Controlled Trial Comparing Neoadjuvant Chemoradiotherapy Plus Surgery Versus Surgery Alone for Oesophageal Cancer (Cross) | J. Bonenkamp - Academia.edu

Accuracy of detecting residual disease after neoadjuvant chemoradiotherapy for esophageal squamous cell carcinoma (preSINO trial): a prospective multicenter diagnostic cohort study | BMC Cancer | Full Text

Patient selection process. nCRT neoadjuvant chemoradiotherapy, dCRT... | Download Scientific Diagram

Nivolumab plus chemoradiotherapy in locally-advanced cervical cancer: the NICOL phase 1 trial | Nature Communications
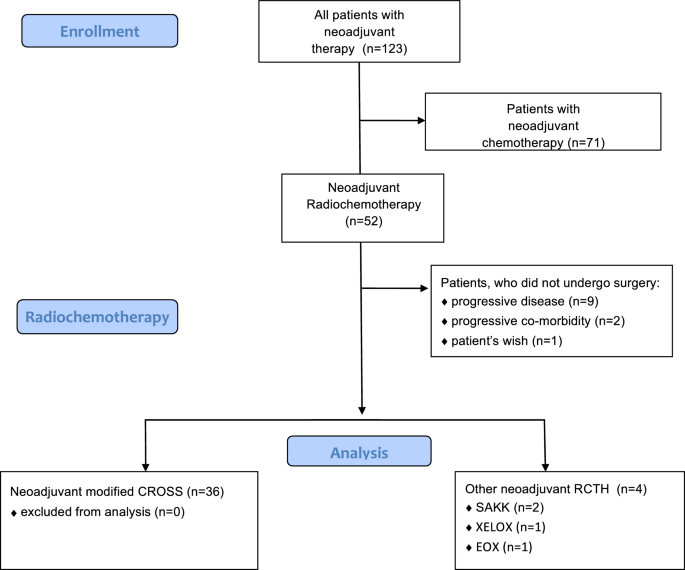
Modification of preoperative radiochemotherapy for esophageal cancer (CROSS protocol) is safe and efficient with no impact on surgical morbidity | Strahlentherapie und Onkologie

Preoperative pembrolizumab combined with chemoradiotherapy for esophageal squamous cell carcinoma: Trial design - ScienceDirect
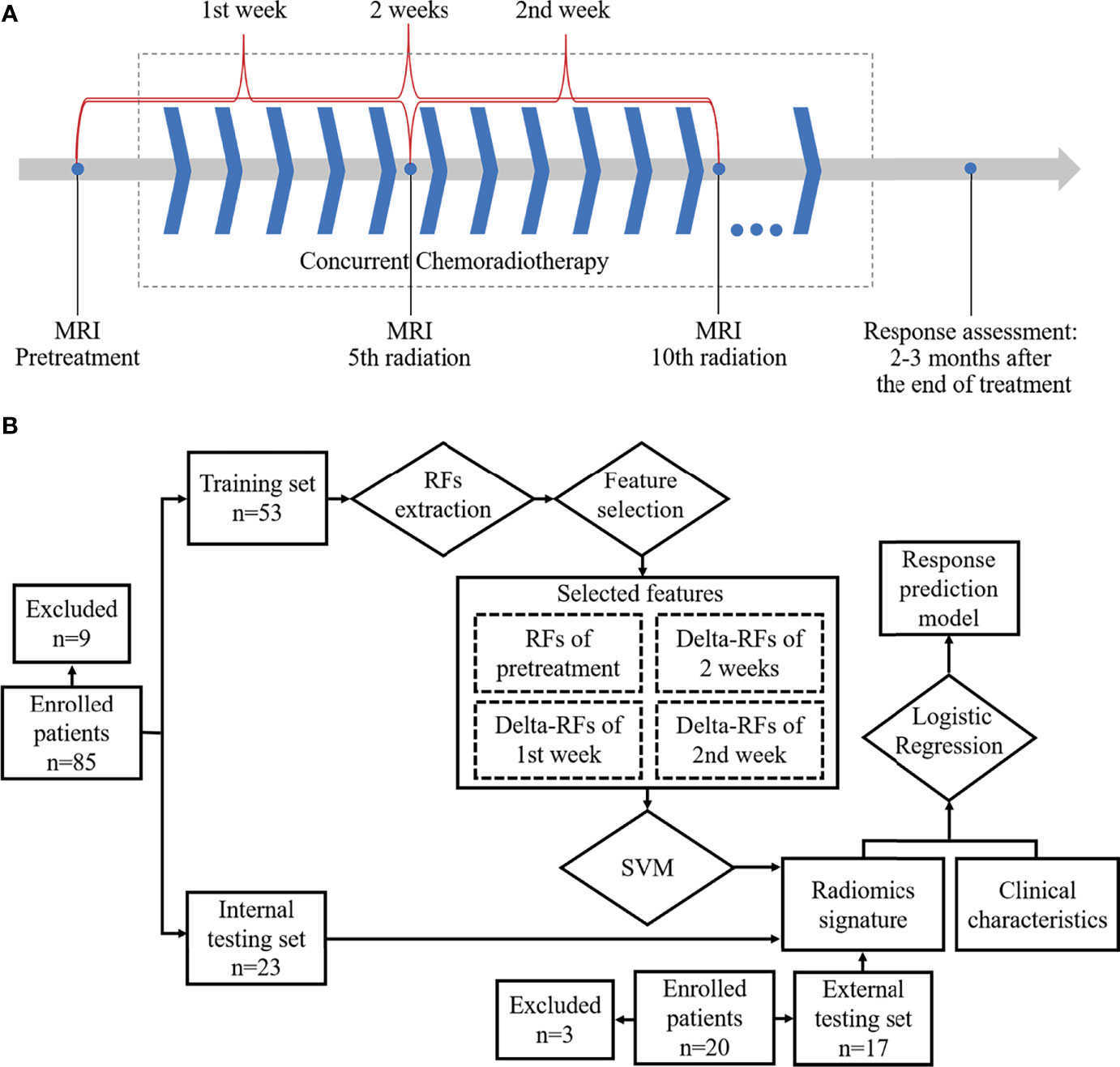
Frontiers | Response Prediction to Concurrent Chemoradiotherapy in Esophageal Squamous Cell Carcinoma Using Delta-Radiomics Based on Sequential Whole-Tumor ADC Map

Yüksel Ürün: KEYNOTE-412 trial shows pembrolizumab + chemoradiotherapy doesn't significantly improve event-free survival in unselected advanced head and neck squamous cell carcinoma vs placebo - OncoDaily

Three-arm phase II trial comparing camrelizumab plus chemotherapy versus camrelizumab plus chemoradiation versus chemoradiation as preoperative treatment for locally advanced esophageal squamous cell carcinoma (NICE-2 Study) | BMC Cancer | Full Text