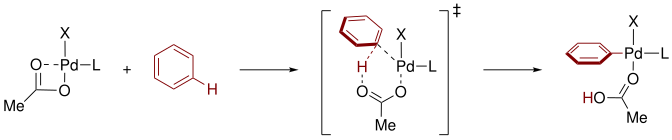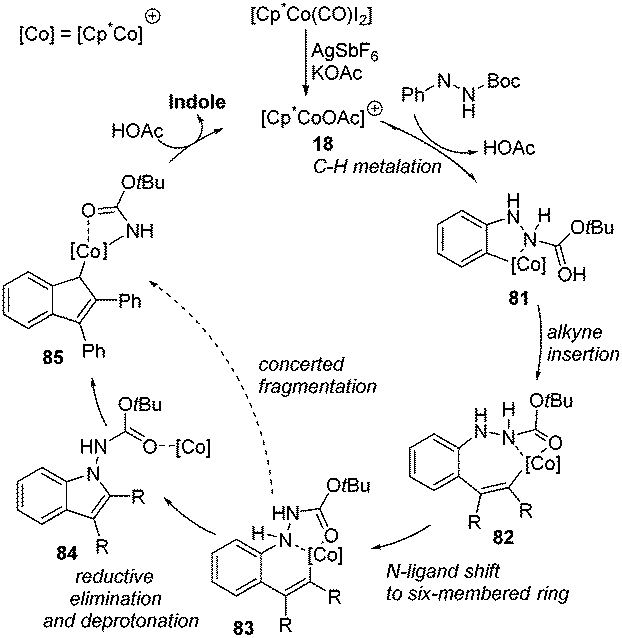
C–H functionalization by high-valent Cp*Co( iii ) catalysis - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC09651D

A generalized representation of (a) concerted metalation–deprotonation... | Download Scientific Diagram
Gibbs free energy profile for the concerted metalation–deprotonation... | Download Scientific Diagram
C–H Activation versus Ring Opening and Inner- versus Outer-Sphere Concerted Metalation–Deprotonation in Rh(III)-Catalyzed Ox

Figure 1 from Investigation of the mechanism of C(sp3)-H bond cleavage in Pd(0)-catalyzed intramolecular alkane arylation adjacent to amides and sulfonamides. | Semantic Scholar

Concerted vs Nonconcerted Metalation–Deprotonation in Orthogonal Direct C–H Arylation of Heterocycles with Halides: A Computational Study - ScienceDirect

Relative Strength of Common Directing Groups in Palladium-Catalyzed Aromatic C−H Activation - ScienceDirect
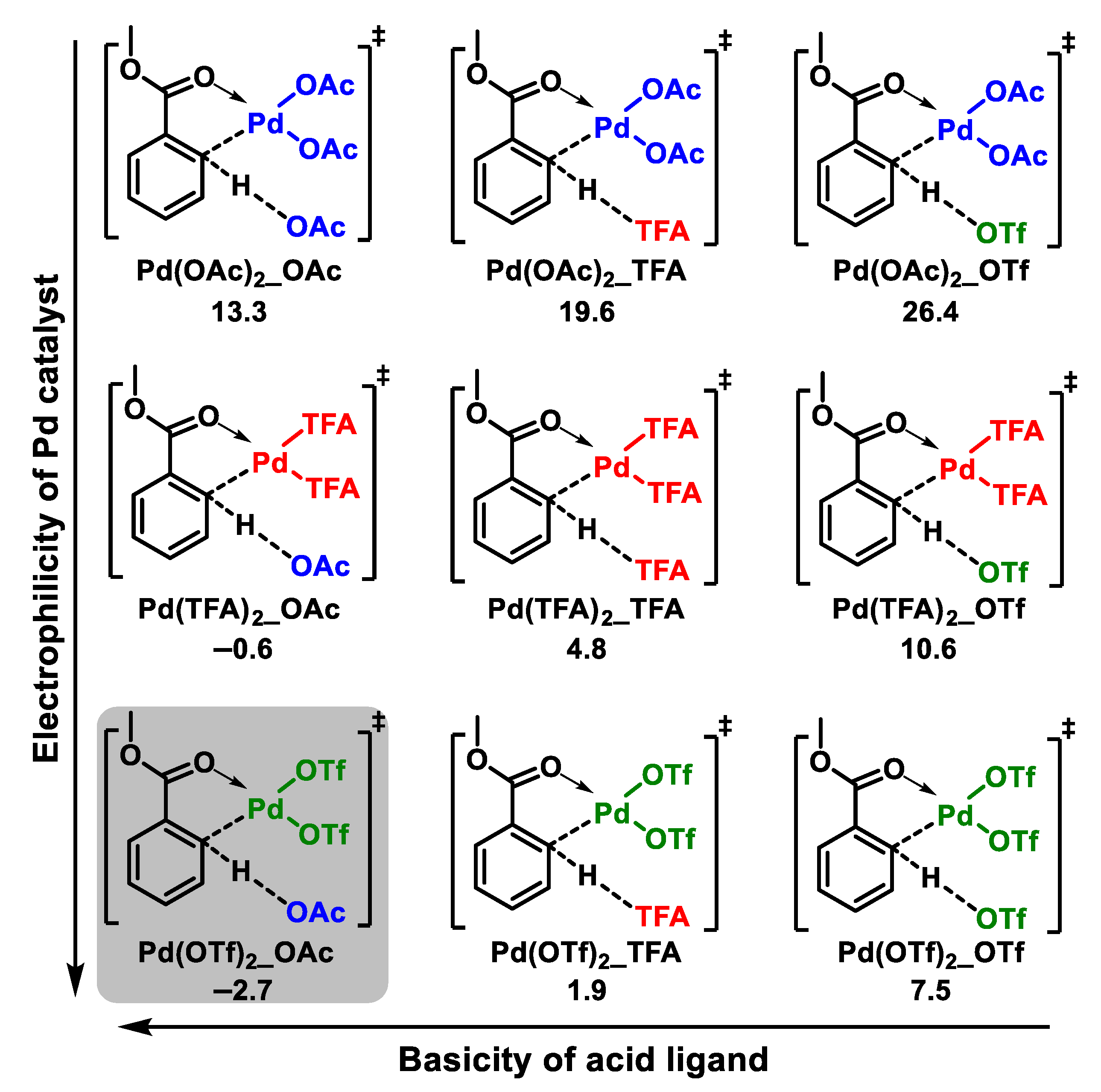
Molecules | Free Full-Text | The Activating Effect of Strong Acid for Pd-Catalyzed Directed C–H Activation by Concerted Metalation-Deprotonation Mechanism

A generalized representation of (a) concerted metalation–deprotonation... | Download Scientific Diagram

Computational exploration of Pd‐catalyzed C–H bond activation reactions - Yang - 2018 - International Journal of Quantum Chemistry - Wiley Online Library

Oligothiophene Synthesis by a Distinct, General C−H Activation Mechanism: Electrophilic Concerted Metalation-Deprotonation (eCMD) | Catalysis | ChemRxiv | Cambridge Open Engage
Analysis of the Concerted Metalation-Deprotonation Mechanism in Palladium-Catalyzed Direct Arylation Across a Broad Range of Aro

Figure 1 from Analysis of the concerted metalation-deprotonation mechanism in palladium-catalyzed direct arylation across a broad range of aromatic substrates. | Semantic Scholar
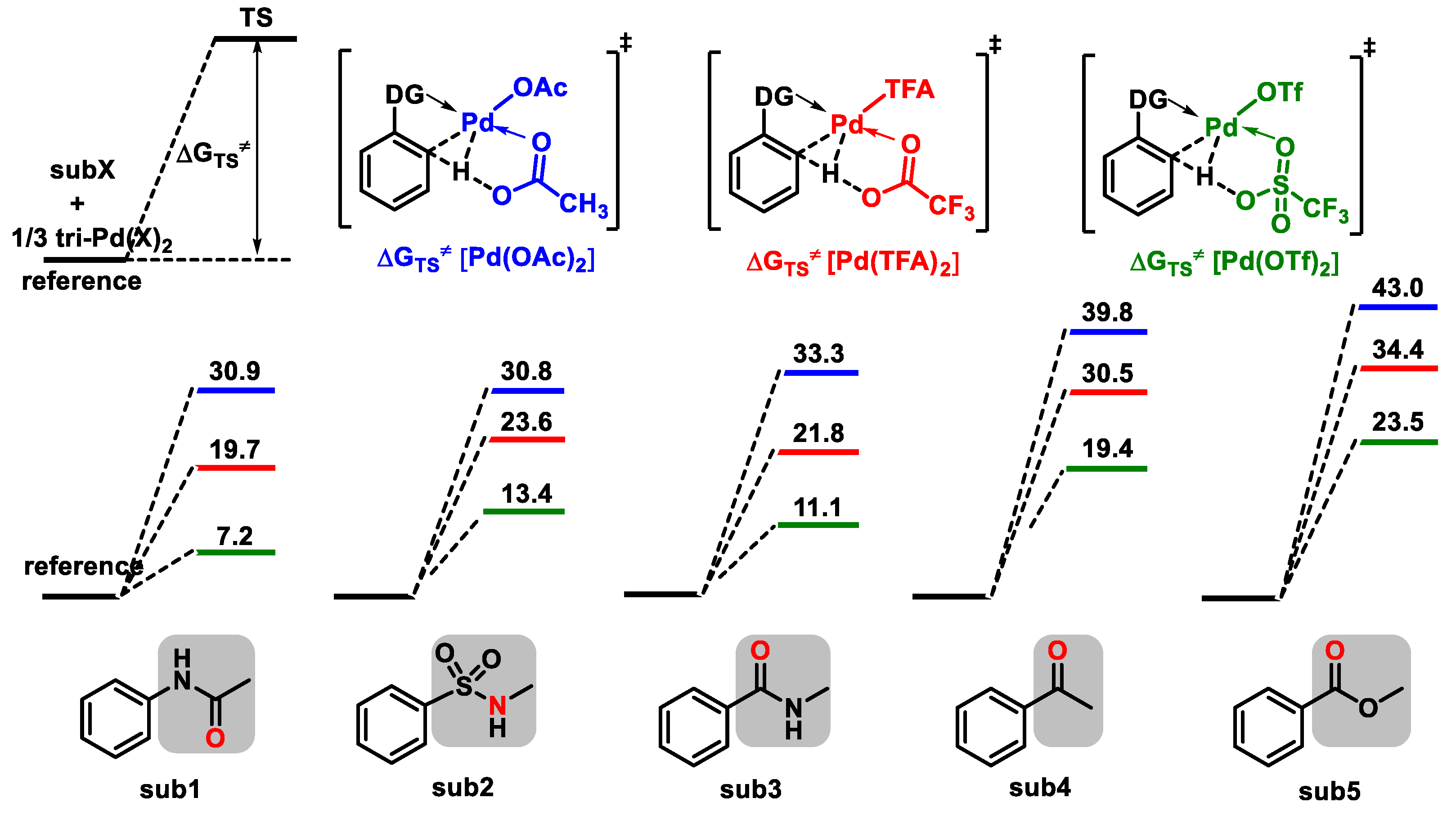
Molecules | Free Full-Text | The Activating Effect of Strong Acid for Pd-Catalyzed Directed C–H Activation by Concerted Metalation-Deprotonation Mechanism

Molecules | Free Full-Text | The Activating Effect of Strong Acid for Pd-Catalyzed Directed C–H Activation by Concerted Metalation-Deprotonation Mechanism
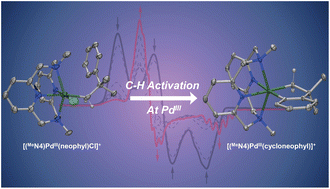
C–H bond activation via concerted metalation–deprotonation at a palladium(iii) center - Chemical Science (RSC Publishing)
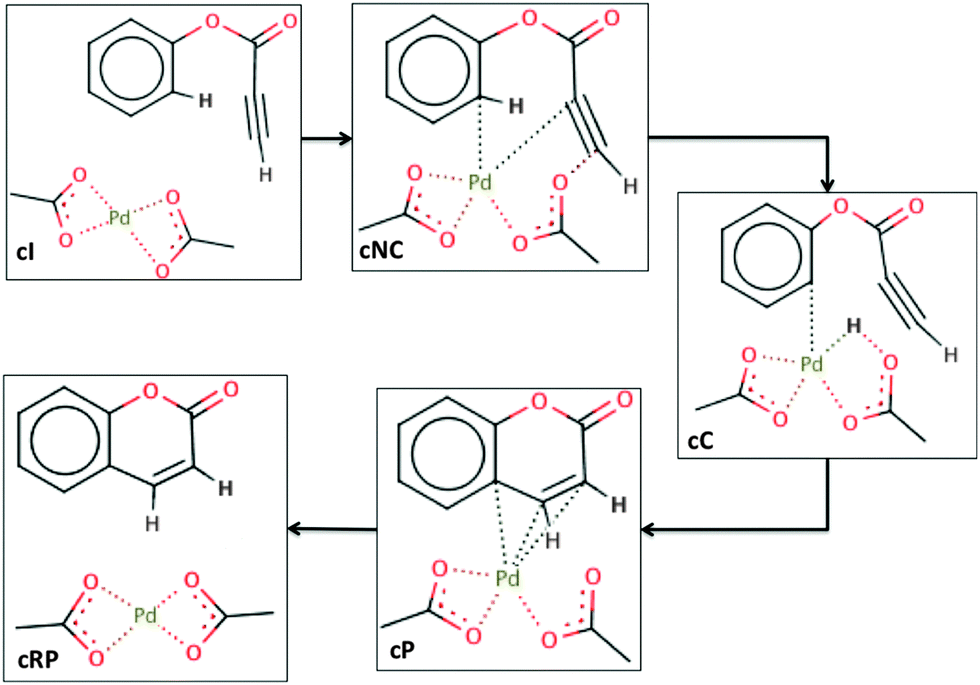
The mechanism of the Pd-catalyzed formation of coumarins: a theoretical study - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C4CP03418J

C-H Bond Activation via Concerted Metalation-Deprotonation at a Palladium(III) Center | Organometallic Chemistry | ChemRxiv | Cambridge Open Engage