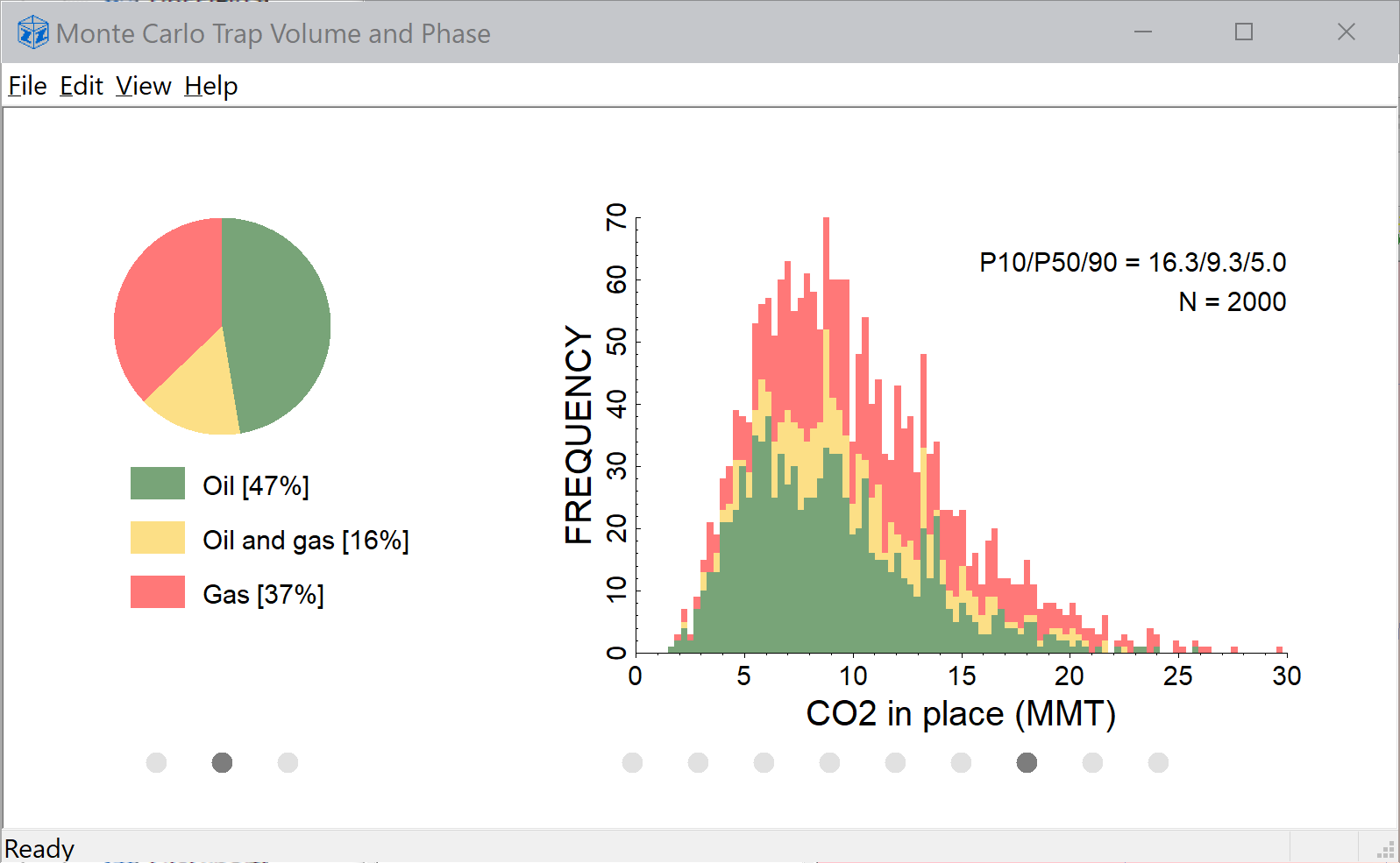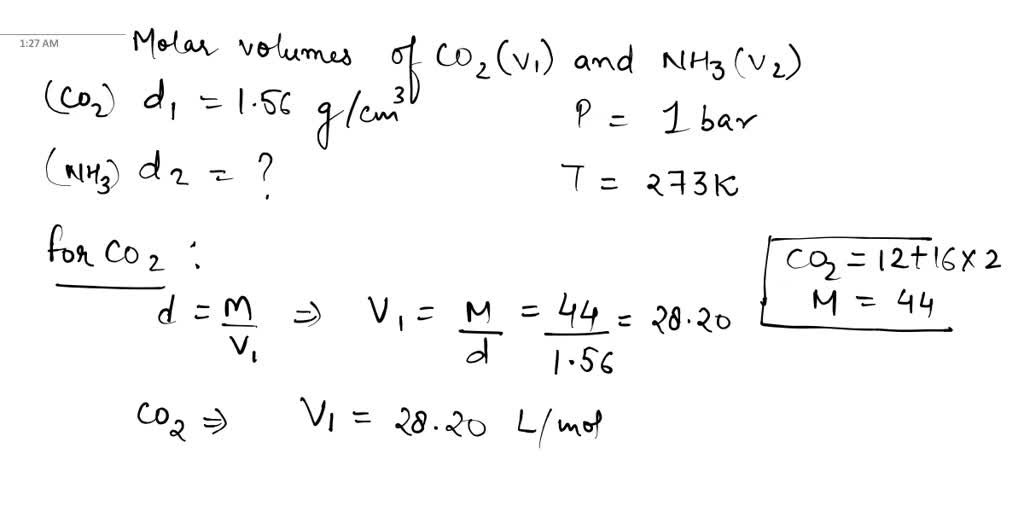
SOLVED: At standard temperature and pressure the molar volumes of CO2 and NH3 gases are 22.31 and 22.40 L, respectively. The density of crystalline CO2 at 160 K is 1.56 g/cm^3. Calculate
Vettoriale Stock Chemistry model molecule carbon dioxide CO2 scientific element formula. Integrated particles natural inorganic 3d molecular structure consisting. Two oxygen and carbon volume atom eps vector spheres | Adobe Stock