The major organic product formed from the following reaction 3 Methylbutan 2 one [ (i)CH3NH2 (ii)LiAlH4 (iii)H2O ] ——> [AIPMT (Prelims) 2005]

Is CH3NH2 polar or non-polar? - Polarity of CH3NH2 | Molecular geometry, Covalent bonding, Electron configuration
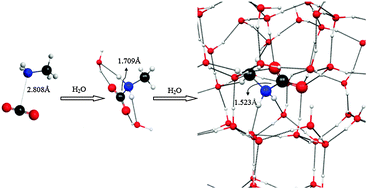
A computational study on the structures of methylamine–carbon dioxide–water clusters: evidence for the barrier free formation of the methylcarbamic acid zwitterion (CH3NH2+COO−) in interstellar water ices - Physical Chemistry Chemical Physics (RSC

Solved) - Methylamine is an organic compound with a formula of CH3NH2. This... (1 Answer) | Transtutors

The energetics and kinetics of the CH3CHO + (CH3)2NH/CH3NH2 reactions catalyzed by a single water molecule in the atmosphere - ScienceDirect
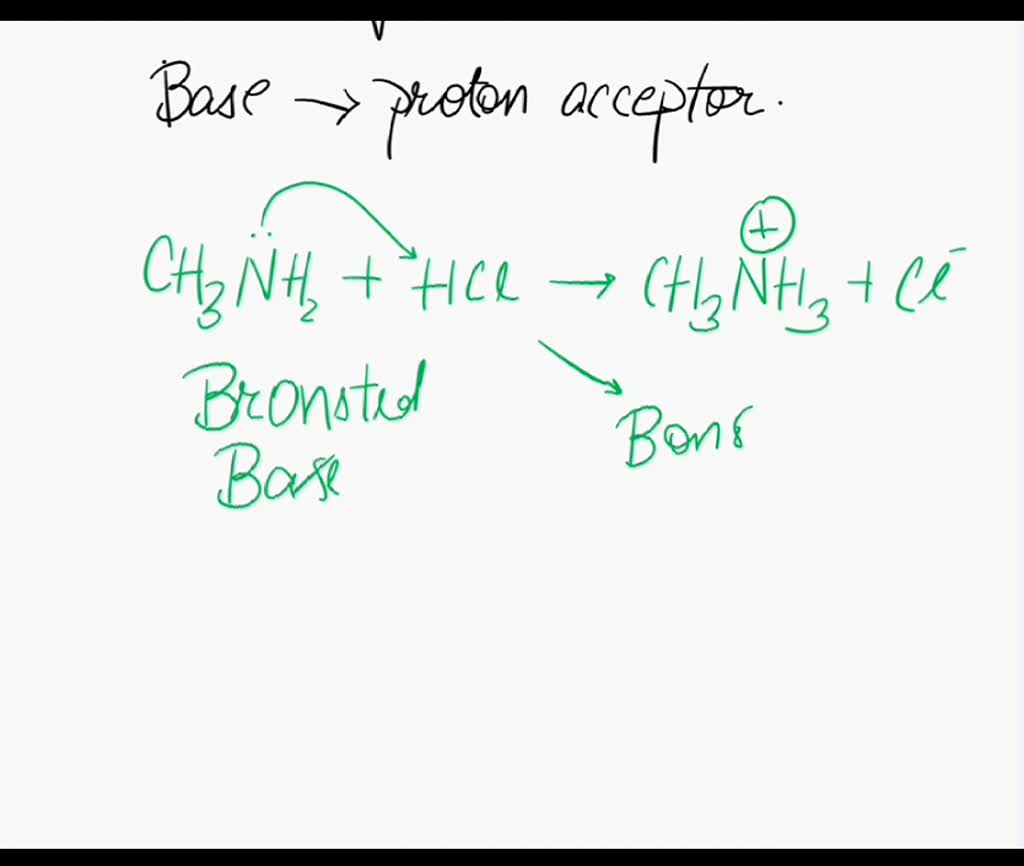


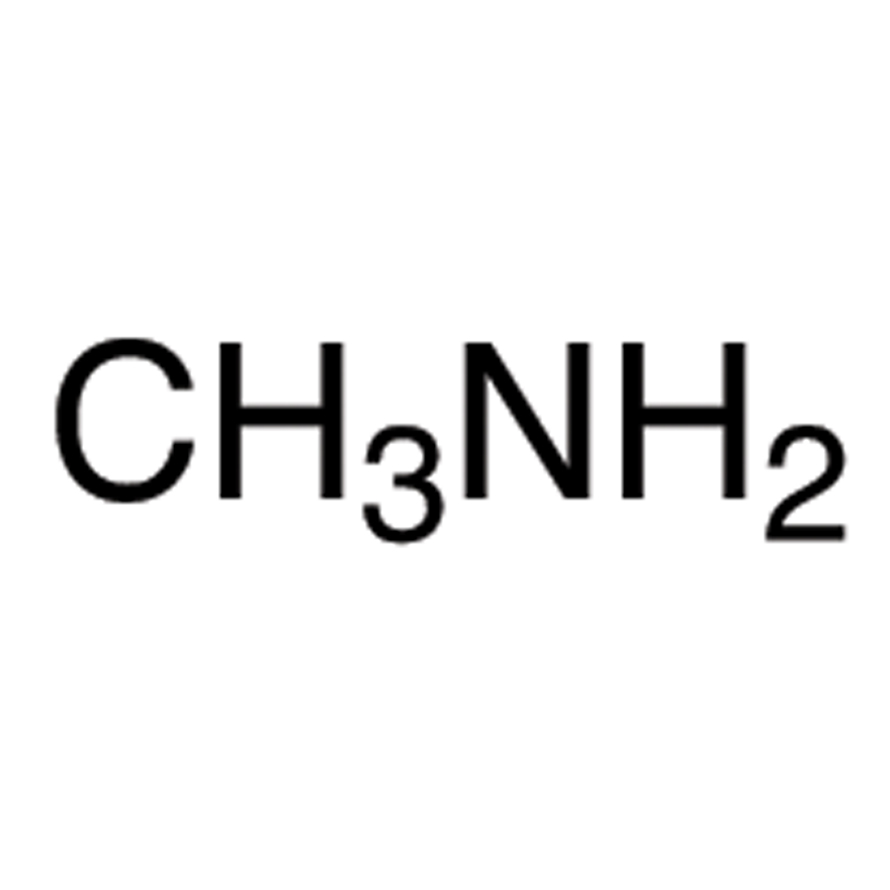

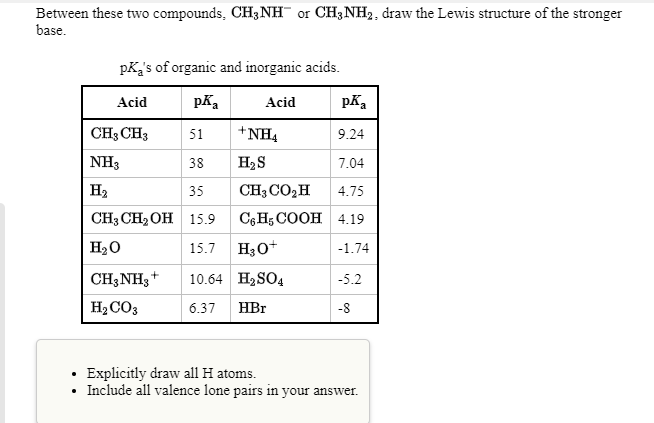



![Solved QUESTION 2 [30 MARKS] Methylamine is an organic | Chegg.com Solved QUESTION 2 [30 MARKS] Methylamine is an organic | Chegg.com](https://media.cheggcdn.com/study/ce5/ce5a6923-d4fb-4f52-946d-c438f9ee3ab9/image.png)


