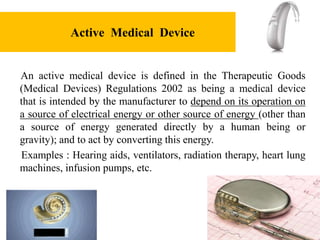
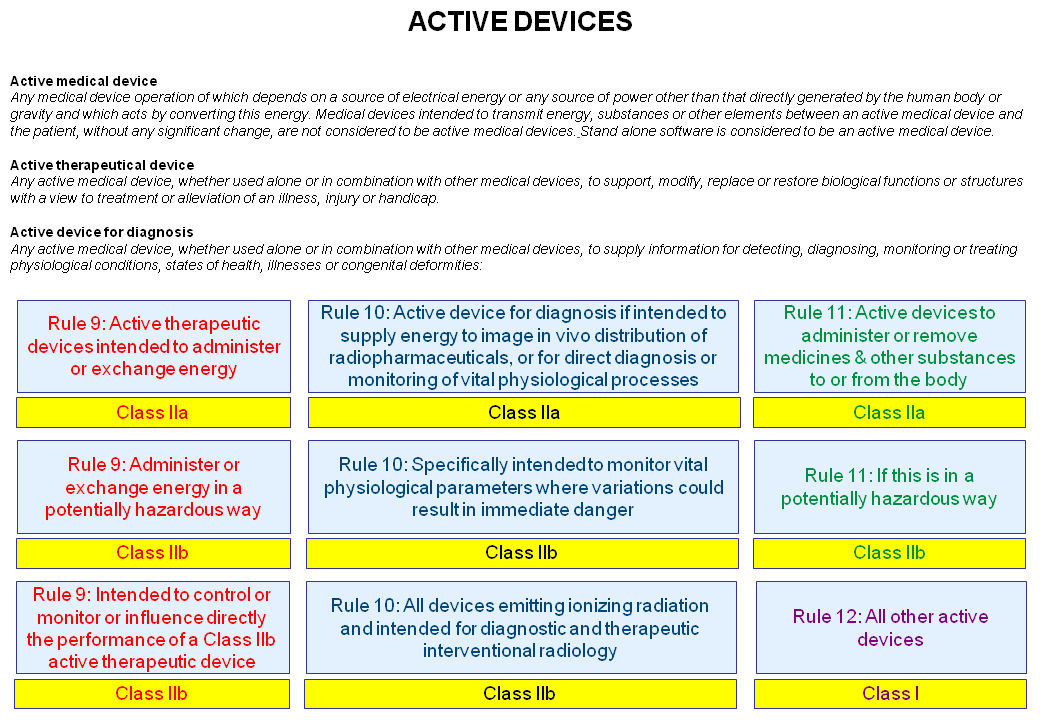
GSPR 12. Requirements for devices incorporating active substances or substances absorbed by or locally dispersed in the human body.
Regulatory Requirements for Medical Devices and IVDs in India Prof. Aseem Sahu Prof. Malay Mitra Department of Biotechnology, Mi

Guidelines for Classification of Medical Devices - CE Marking (CE Mark) for Medical Devices - EU Council Directive 93/42/EEC